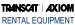|
|
|||||||||
| Software: RF Cascade Workbook | RF Symbols for Office | RF Symbols & Stencils for Visio | Espresso Workbook | ||||||||||
|
|||||||||||||||||||||||||||||||
   |
   |
||||||||||||||||||||||||||||||
|
Please Support RF Cafe by purchasing my ridiculously low-priced products, all of which I created. RF & Electronics Symbols for Visio RF & Electronics Symbols for Office RF & Electronics Stencils for Visio T-Shirts, Mugs, Cups, Ball Caps, Mouse Pads These Are Available for Free |
|||||||||||||||||||||||||||||||

Crystal Finishing
November 1944 Radio News
Article
Radio-Electronic Engineering Department (insert)

| November 1944 Radio News |
 [Table of Contents] [Table of Contents]
Wax nostalgic about and learn from the history of early electronics. See articles from Radio & Television News, published 1919-1959. All copyrights hereby acknowledged. Special Insert: Radio-Electronic Engineering Department |
With a last name like Blattenberger and an unusually spelled first (Kirt), I learned a long time ago not to poke fun at anybody's name, but surely the parents of Dr. Hal F. Fruth had a sense of humor when assigning their little bundle of joy in such a manner (HalF Truth?). Maybe I assume too much. Anyway, the good doctor published in this issue of Radio News magazine a very extensive treatise on the art and science of mass producing radio frequency crystals at a time when the country - and free world - was in urgent need of them. He notes that, "Prior to Pearl Harbor Day, the world production of these plates and bars could be counted by the thousands. The present production rate has skyrocketed more than one hundred fold so that the present production rate is nearly 30,000,000 units per year." That was in late 1944. World War II was the epitome of proof for the old adage about necessity being the mother of invention. The global crystal oscillator market in 2017 was $2.64B and forecasts predict $3.69B by 2025. Divide those number by any average cost you please and it represents a huge amount of crystal oscillators.
Crystal Finishing

Dr. Hal F. Fruth
The author, with his colleagues, originated and perfected the mill-finishing methods discussed here.
Centrifugal beveling machine for beveling and pre-dimensioning crystals after lapping.
Raw quartz, finished crystal blank, and complete crystal unit.
By Dr. Hal F. Fruth
Assistant Chief Engineer, Research Division, Galvin Manufacturing Company
A discussion of various methods of finishing crystals with particular emphasis on mill finishing methods including milling time and abrasives.
The piezoelectric crystal is a crystal that is responsive to electric and mechanical pressures. In its simplest concept, mechanical pressure along one axis gives rise to dimensional changes and electrical charges on faces perpendicular to the other axis, and vice versa. This piezoelectric effects finds many uses. This discussion will be confined to electric oscillators and filters and generally to quartz oscillator and filter plates and bars.
Prior to Pearl Harbor Day, the world production of these plates and bars could be counted by the thousands. The present production rate has skyrocketed more than one hundred fold so that the present production rate is nearly 30,000,000 units per year. Coupled with this has been a large quality increase and a production cost reduction, quality for quality, to about one tenth of previous costs. This accomplishment ranks among the highest of American research, development and production miracles.
The most critical and precise step in crystal production is that of "crystal finishing," i.e., the last and final operations on the crystal blank to bring it up to the best activity and the precise frequency, and still produce a surface of greatest stability. Much has been said about modern precision and "super finish." High frequency crystal blanks have the smallest thickness tolerance of any existing manufactured article. In order to finish a BT-cut, 10 megacycle crystal to a tolerance of ±100 cycles one would have to grind it to ±1 Angstrom unit or ± 4 X 10-9 inches. Stating it another way, ±100 cycles = ±0.01 microinches. The thickness tolerance is equivalent to a very small fraction of a wave length of blue light and is nearly equal to a layer of quartz one molecule thick.
Finishing Methods - Hand Finishing
A bank of commercial milling machines for finishing and cleaning crystals to final frequency.
Close-up view of a two-jar machine.
Ten-jar milling machine, with all jars operating from a single drive.
Fig. 1 - Relative activity plotted against beveling time for a typical crystal.
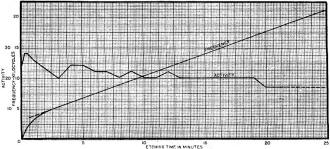
Fig. 2 - Frequency change plotted against etching time for a 10 mc. crystal lapped with No. 600 silicon carbide. milled 20 kc., and then etched. Relative activity also shown.
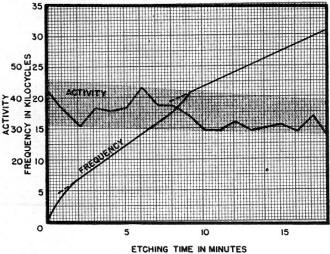
Fig. 3 - Frequency change plotted against milling time for a 5.1 mc. crystal. The relative activity and the change in curvature of the crystal are also indicated.
Fig. 4 - Plot of frequency change vs. etching time for an 8599 kc. crystal lapped with No. 600 abrasive and then finished by etching. Relative activity is also shown.
Fig. 5 - Plot of frequency change and relative activity vs. etching time for a 10,000 kc. crystal lapped with No. 600 abrasive. Break in curve shows removal of surface layer.
Fig. 6 - Plot of frequency change and relative activity vs. milling time for an 8170 kc. crystal lapped with No. 600 abrasive and milled with No. 600. garnet, alcohol and water.
Fig. 7 - Average frequency increase vs. milling time for 8000 kc. crystals milled with quartz, garnet, and Haystellite. 4 to 8 mesh. plus 6% by volume of No. 600 silicon carbide.
Fig. 8 - Average frequency increase VB. milling time for various crystals, milled with 4 to 8 mesh Haystellite plus 6% by volume of No. 600 silicon carbide.
Fig. 9 - Frequency increase vs. milling lime for several individual crystals milled with 4 to 8 mesh Haystellite plus 6% by volume of No. 600 silicon carbide.
Fig. 10 - Average frequency change and relative activity plotted against milling time for 6230.4 kc. crystals milled in garnet. Inset shows typical individual activity curves.
Fig. 11 - Average frequency increase for crystals milled with 4 to 8 mesh garnet and No. 600 silicon carbide, 6% by vol.; 55 r.p.m.
Fig. 12 - Frequency increase per hour as a function of granule density for 8 mc. crystal. Granules 4 to 8 mesh, plus 6% of No. 600 SiC.
Fig. 13 - Effect of milling on contour of a 4750 kc. crystal. Contour change for 3 mc. crystal. 0.0002 inch/100 kc. change: 5 mc. 0. 0001 inch/100 kc.: and 8 mc. ... 0.00006 inch/100 kc.
Fig. 14 - Frequency increase vs. etching time for an 8000 kc. BT crystal etched in 25% "Frequency-Etch" solution.
A few years ago the most generally used method employed for finishing crystals to final frequency and activity was to rub the crystal by hand over a flat glass surface coated with a water suspension of very fine emery. A figure-eight motion was usually used and this grinding repeated on both sides a number of times, the crystal being washed between measurements. This procedure was repeated a large number of times coupled with grinding and beveling of the edges until the best activity and proper frequency was obtained. This procedure required highly skilled "finishers" who could produce from 30 to 60 crystals per day. Much variation and many uncertainties were coupled with this method, and, in the light of modern findings, a poorer quality and less stable crystal and crystal surface was produced. At times, light mechanical pressure methods were coupled with or substituted for this hand finishing process with no better results. Even very accurate lapping machines using very light plates, precise motion and very fine abrasives did not produce a really stable surface.
The surface produced by the so-called "pressure" methods, in spite of its good appearance under the microscope, was covered with minute cracks, fissures, scratches, and pits, as well as deeper scratches, often made invisible by being packed full of a nearly transparent disoriented quartz abrasive, metal and glass, which was very difficult to remove by washing or scrubbing. However, this foreign material could be detected by X-ray and could be removed by etching or by exposure to high humidity temperature and time. This change in the surface of crystals is considered to be the principal cause of their instability.
Etching
A few manufacturers have for many years finished crystals by etching. The usual etchant was formerly hydrofluoric acid, with all its hazards and uncertainties. Recently a number of safer etchants have appeared on the market in the form of liquids, pastes, or salts, most of them containing a fluoride salt. This method, when used with good control, is faster and cheaper than hand finishing, lends itself to semi-automatic mechanical methods and can produce high quality surfaces and crystals when not carried too far. This method, must, however, be preceded by very careful grinding methods or processes that produce good smooth surfaces so that the etchant will give predictable frequency changes and will be prevented from greatly exaggerating cracks, scratches, etc., which would result in inferior quality crystals, greater fragility, less stable frequency, and lowered activity. (Figs. 4 and 5.)
Other Finishing Methods
A few laboratories and factories have brought about frequency changes by such methods as X-ray, heat treatment, and vibration in or on abrasive materials, sand-blasting, and falling or flowing abrasive on the surface of the crystal. None of these methods has found extensive use in production.
Mill Finishing or Tumbling
Realizing that the above methods were accompanied by uncertainties and high cost, and produced inferior surfaces, extensive experiments were undertaken to find a method for removing quartz from the surfaces of crystals which would be gentle, random, non-chemical, fast, controllable, "non-custom," predictable and suitable for mass production.
Such a method seemed to be suggested by a quotation from OVID (43 A.D.). Gutta cavat lapidem, non vi sed saepe cadendo, "Not by strength but by constant falling does the drop hollow out the stone."
It was found that crystals can be put into a pebble or tumbling mill with rather coarse (4 to 15 mesh) granules and an abrasive powder, and tumbled in a tumbling jar without breaking or injuring the crystals even when the jar is rotated at speeds up to 60 rpm. Furthermore, the frequency change is linear with time, and is predictable.
Photographs of crystals finished by various methods, showing conditions of crystal edges. Left, crystals hand beveled, center, crystals as they come from lapping and edge grinding machine. and right, crystals machine finished in the beveling machine.
Table I - Frequency changes of four lots of crystals after various treatments.
Certain precautions should, of course, be taken and certain variables must be controlled. It was found that the most important variables were:
(1) Shape and size of the pebble mill, or tumbling container, and the manner of its rotation.
(2) Speed of rotation.
(3) Size and density of the granules.
(4) Percent of filling of the jar.
(5) Liquid medium and wetting agent.
1. Extensive experiments on the action of the pebble mill showed that in order to control the thickness of the crystals and not to disturb the outer periphery and the contour in too large a degree, it was necessary to adjust the charge, the shape of the vessel, and the angle and speed of rotation, in such a manner that. the charge would be lifted at an angle without excessive spilling over the upper surface and would slide down at a point when the angle of repose reaches the critical value. At that angle, the upper layer slides down in this laver, producing the grinding action. The crystals would slide over the heavier granules on the top layer and would experience a relative motion parallel to the faces of the crystals, in a very highly random motion but under very low pressure. If such a set-up is made, the contour and the speed of cutting are controllable and predictable.
2. It was found that the rate of change of frequency varied in a nearly linear manner with the r.p.m., and that a tumbling mill or jar approximately 6" in diameter having four flat sides and rotating between 10 and 55 r.p.m. gave the best results. At higher speeds, the crystals, might be injured by being thrown against the sides too severely, and the cutting action would be cut down.
3. It appeared early in the engineering work that it. was desirable to have two types of materials with the crystals in the mills, one of them to give weight and be the force factor, "granules," and the other to do the cutting, a hard, rather fine material, e.g., 600 silicon carbide. It was found that the rate of frequency change varied directly with the density of the granules (Fig. 12). However, the densest material used, 4-8 mesh Haystellite, was too heavy for glass jars and was not suitable in general for crystals above 7 mc. Using a suitable density material for various types of crystals, it was found that the cutting rate could be varied with the amount of grinding necessary for the crystal in question and with the delicacy of the wafer or blank.
4. It was found that the amount of charge of the jar was significant; too small a quantity was not feasible and too large a quantity reduced the cutting rate. It appeared that a jar five-eighths full of solids gave about the best performance.
5. In order to obtain uniform cutting and obtain clean crystals at all stages, it was found desirable to add a wetting agent, soap or detergent, such as aerosol "Alconox" or some soap which would help to put the fine particles into colloidal suspension, so that they could be washed away.
It is of utmost importance that no pockets or crevices be present in which crystals might temporarily be lodged and that no flat surfaces exist, against which they might be held in any part of the interior of the vessel, in order to assure that all crystals will receive equal treatment during the entire milling time.
The size of the abrasive seems to influence the cutting rate and the quality of the surface produced in some degree. However, any silicon carbide or aluminum oxide in grit sizes from 320 to 800 is suitable for the purpose. The optimum quantity is usually between four and eight percent by volume of the solid content in the container. It is desirable to add or renew the abrasive charge every 5 hours, if the cutting rate is to remain unchanged over many hours.
By omitting abrasive material from the charge and using the granules only, together with water and a wetting agent and using granule sizes between 10 and 25 mesh, it was found that crystals could be cleaned very effectively. Tests revealed that in this manner a very high quality and stable surface could be produced, without the use of any solvent other than hot distilled water.
Commercial Application
As seen from the graphs (Figs. 6, 7, 8, 9, 11 and 12) the variation of frequency vs. time is essentially a straight line function, and since the individual crystals will behave in very nearly the same manner (Fig. 9), the process lends itself readily to mass production.
A number of procedures may be used; one of them is to take crystals from the lapping machines, ranging from 100 to 120 kc. away from final frequency, and sort them into groups of plus and minus 5 kc. each. By referring to the charts for the frequency in question and the granule and abrasive charge, one can easily estimate the number of hours necessary to bring the farthest one to the frequency which is estimated to be from 5-10 kc. away from final frequency.
The other groups are then added in suitable intervals until all groups have been added, and the mills stopped when it is estimated that all of them are from 5 to 10 kc. short of final frequency. These are now channeled into groups plus and minus 1 kc. and the procedure continued for final adjustment. It is usually desirable to allow 1 to 2 kc. tolerance for final washing as described above.
Another procedure would be to time each group of crystals in a separate mill and stop the machines by means of a clock at a time when they arc from 5-10 kc. away from final frequency.
Contour
It is the belief of many crystal manufacturers that the radius of curvature of the surface of the crystal, flatness or contour, is of vital importance in the control of the quality, activity, temperature stability, and harmonic content of the crystal blank. It was learned, contrary to expectations, that in mill finishing crystals to frequency there was a continued change in contour, the crystals becoming more and more flat. Fig. 3 and other experiments indicate that a contour or convexity of 0.0002" to 0.00005" is more desirable than absolute flatness or a larger convexity. It is thus possible to allow crystals to become more and more convex during the lapping processes on lapping machines such as the so-called Hoffman or Atlas laps, and then allowing the milling process to remove enough quartz to reduce the contour to the desirable height.
Pre-Dimensioning
Many crystals, particularly those of the BT or AT variety, have to be manufactured with very great care, surface condition and surface cleanliness being of utmost importance in insuring high activity and stability with time, temperature and humidity. The activity at room temperature and the active range of minus 40°C to plus 90°C is active throughout a required temperature function of the dimensions such as length, width and thickness. These dimensional relationships must be maintained to very close tolerances. However, in the case of crystal frequencies between 4 and 10 mc. these dimensional requirements are so very exact that it would be practically impossible to know or maintain them commercially. The random method is therefore employed. This consists of taking crystals from the mills or from the tumbling machines, testing them for activity at room temperature and throughout the temperature range, selecting the satisfactory ones and returning the others for more milling or beveling. Figs. 1 and 10 indicate the type of curve an individual crystal will follow when its dimensions are changed very slightly by beveling or by changing its frequency by milling or other processes. When rejected crystals have been so treated there is at least a 50% chance that they will be satisfactory. Those that are not yet satisfactory may receive additional treatment. If the angle of the crystal was correct and the quartz of proper quality, it is seldom that less than 95% of the crystals will not eventually respond to this treatment.
In the types of crystals mentioned above, surface conditions and surface cleanliness are of the utmost importance in order to insure frequency and activity stability over a long period of time and through a large range of conditions. This surface must not only be clean but it must be free from imbedded materials pressed into the surface; it must also be free from minute cracks and injured or disoriented quartz. There are many methods such as profilometer, electron microscope, and X-ray methods of determining the depth of this undesirable layer, sometimes referred to as the Beilby layer. Another method for determining the depth of this layer is to plot frequency change vs. etching time and note a sharp change of curvature or an abrupt lowering of the etching rate (Figs. 4, 5 and 14). The point of the beginning of linearity usually indicates a time when most of the surface "rubble" has been removed. It was found desirable to etch the crystal to a point where the straight line function begins. It is to be noted that in these curves, this point lies between 10 and 25 kc. in one case, and as much as 300 in Fig. 14. This point of beginning of linearity can be very greatly lowered by proper treatment of the crystal previous to etching.
It has also been observed that there is a general downward trend of activity with etching time when the surface prior to etching has not been properly prepared (Fig. 2). It is to be noted there is no downward trend of activity with milling time even if the crystal has been moved as much as 200 kc. (Fig.10).
In order to determine the life quality of crystals, four varieties of crystals were prepared and tested, and the results are given in the aging tests (Table I).
Listed are aging results of four lots crystals that were finished to final frequency in four different manners. They were assembled and placed in a fungus "Tropical Swamp" humidity chamber for 60 days. The humidity chamber had a temperature of 80° to 100° F. and 90% to 100% humidity, changed hourly.
Lot #1 crystals were etched 20 kc. to final frequency 8130 kc.
Lot #2 crystals were milled 20 kc. etched 5 kc. to final freq. 8130 kc.
Lot #3 crystals were milled 20 kc. and mill washed 5 kc. to final frequency 8130 kc.
Lot #4 crystals were milled 20 kc. and etched 20 kc. to final frequency 10,000 kc.
It appears from the tabulations that I lot #3 shows the most desirable stability. The negative changes of many of the groups are probably due to volatile depositions from the phenolic holder and other foreign matter that had accumulated during the tests. It is also possible that an etched crystal whose surface was not properly prepared before hand had etching compounds entrapped in microscopic pits and fissures which could not be washed out or removed. This could continue to weaken the crystal without removing materials. Such frequency lowering has been encountered in spite of great care to keep crystals very clean.
There is much evidence that quartz oscillator crystals can be finished by the milling method safely and economically, and that the method produces crystals of equal or superior quality to those manufactured by any other finishing method. It is particularly superior in all respects to hand-finishing or any other high pressure method. The method also lends itself very well to combined finishing and beveling methods and as a preparatory grinding and surface stabilizer prior to etching or in etch-cleaning processes. Literally many millions of crystals have been treated and finished by this method or in combination with other methods and are giving an excellent account of themselves in life tests and in actual service under the severest service conditions.

Copyright: 1996 - 2026 |
About RF Cafe RF Cafe began life in 1996 as "RF Tools" in an AOL screen name web space totaling 2 MB. Its primary purpose was to provide me with ready access to commonly needed formulas and reference material while performing my work as an RF system and circuit design engineer. The World Wide Web (Internet) was largely an unknown entity at the time and bandwidth was a scarce commodity. Dial-up modems blazed along at 14.4 kbps while tying up your telephone line, and a lady's voice announced "You've Got Mail" when a new message arrived... |
Copyright 1996 - 2026 All trademarks, copyrights, patents, and other rights of ownership to images
and text used on the RF Cafe website are hereby acknowledge My Hobby Website: My Daughter's Website: |