|
|
|||||||||
| Software: RF Cascade Workbook | RF Symbols for Office | RF Symbols & Stencils for Visio | Espresso Workbook | ||||||||||
|
|||||||||||||||||||||||||||||||
   |
   |
||||||||||||||||||||||||||||||
|
Please Support RF Cafe by purchasing my ridiculously low-priced products, all of which I created. RF & Electronics Symbols for Visio RF & Electronics Symbols for Office RF & Electronics Stencils for Visio T-Shirts, Mugs, Cups, Ball Caps, Mouse Pads These Are Available for Free |
|||||||||||||||||||||||||||||||

Light Emitting Diodes
November 1970 Popular
Electronics

November 1970 Popular Electronics  Table of Contents Table of Contents
Wax nostalgic about and learn from the history of early electronics. See articles from Popular Electronics, published October 1954 - April 1985. All copyrights are hereby acknowledged. |
"One of the least known but most fascinating of semiconductor devices is the light-emitting diode (LED). Until quite recently, these devices were too expensive for widespread use." That statement is hard to imagine in 2023. It was written 50 years ago by Mr. Forest Mims, in this November 1970 issue of Popular Electronics magazine. Now, LEDs seem to be in every consumer device, whether it be a simple power ON indicator light or an array of alpha-numeric displays. TV and stereo remotes use infrared LEDs, opt isolators that use LEDs are integral components of modems, motor controllers, and motion sensors. Commercial truck and some passenger vehicle tail lights are made of LED clusters, as are the very expensive LED bulbs that are quickly replacing incandescent and, thankfully, the toxic (mercury) CFL household bulbs. What was once rare is now a commodity - it happens all the time.
Light Emitting Diodes - New Semiconductors for Readout and Communication
 Cover story by Forrest M. Mims, III
Cover story by Forrest M. Mims, III
One of the least known but most fascinating of semiconductor devices is the light-emitting diode (LED). Until quite recently, these devices were too expensive for widespread use; however, technological advances in their fabrication now make possible moderately low prices so that they are attractive to the electronics experimenter.
The first recorded instance of light being generated by a "diode" was in 1907 when H. J. Round touched a pair of battery wires to a crystal of silicon carbide. Much to his surprise, flashes of yellow light were emitted at the contact region of one of the battery wires - he had accidentally discovered the LED. Unfortunately, his discovery was forgotten and not until the early 1950's did scientists once again study semiconductor light emission. At that time several patents were applied for covering LED's made from silicon or germanium - common semiconductor materials. One of these patents not only described the principle of the LED but listed several fascinating uses for the device. Among them were light-beam communication systems, light "radar", and light beam alignment devices.
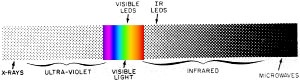
Fig. 1 - Most infrared LED's emit light at a wavelength of about 0.9 microns of the electromagnetic spectrum. Visible LED's may emit from 0.53 microns to the limit of visible region at about 0.75 microns.
Since the LED has an almost unlimited lifetime and because of its low operating current, many claims were made for its potential in flat screen television, indicator lamps, night lights, and even as a source of room lighting. The extremely fast modulation capability of the LED made possible several demonstrations of voice communications permitting two parties to converse over a beam of invisible infrared light for clear weather distances of several miles.
How Does It Do It?
The LED is different from conventional incandescent lamps because the latter give off light as a byproduct of heat. That is, the filament must first be heated to incandescence before light is produced. This, of course, is the reason for placing incandescent filaments in evacuated or gas-filled glass bulbs. If the filament were exposed to oxygen, it would quickly be consumed. Not so with LED's; they operate with or without the presence of air. In fact, the main reason LED's are placed in containers is to protect the rather delicate contact wires. Often, in fact, the LED is simply protected by a layer of clear epoxy which serves both as a protective cover and a lens. Since the LED produces far more light than heat, it is a more efficient source of light than the incandescent lamp.
Production of light by an LED comes under the broad heading of "electro-luminescence." Luminescence describes light produced by means other than incandescence. Since luminescing bodies are generally at the temperature of their environment, they are sometimes referred to as sources of "cold light." Fireflies, many species of fish, rotting wood, marsh gas, and many other objects are sources of luminescent light.
Light from an LED is the result of stimulation by a small electric current. As is the case with all luminescent bodies, the light is a result of sub-atomic events. The cause of these events may be chemical, as with the firefly, or electrical as with the LED.
Light Generation
In the LED, emission of light is called "PN junction luminescence." As shown in Fig. 2, light is emitted at the junction of an LED when electrons which have been stimulated to higher than normal energy levels move across the junction and fall back into their normal places. The energy loss resulting when an electron reoccupies its normal slot in an atom is accompanied by the emission of a photon of light at a wavelength related to the difference in energy between the two bands. Since junction luminescence involves the combination of electrons with holes, physicists often refer to the effect as "recombination radiation."
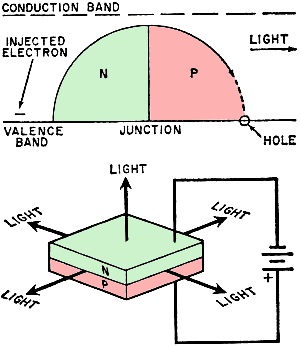
Fig. 2 - Light is emitted when an electron crosses the np junction interface and falls from the excited to the unexcited state.
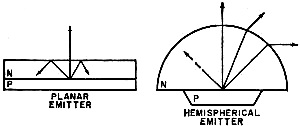
Fig. 3 - The critical interface problem. Much more light is emitted by hemispheric diode but some is lost in the thick n-region. Some planar emitters are provided with a reflector to give greater light output.
1. Internal absorption of light within the p or n region of the semiconductor following its emission at the junction.
2. Reabsorption of light which is reflected back into the diode due to the "critical interface" problem.
3. Resistance at the electrical contacts and in the semiconductor.
Many of these inefficiencies have been partially resolved as a result of years of research. Consider, for example, the critical interface problem. Since most semiconductors have a high index of refraction, they tend to reflect light at air-semiconductor interfaces. The effect is similar to that of an observer looking through a glass window and seeing his own reflection as well as objects on the other side of the window. The effect results in the loss of light that is generated at the junction.
An ingenious solution to the problem was suggested by early research and was first used commercially by Texas Instruments. It consists of forming the light emitting region of the diode into a dome. Since the light is only bent and not reflected back into the diode, all light reaching the surface is emitted (see Fig. 3). Unfortunately, the dome approach does have its drawbacks - the longer path that the light must travel before being emitted causes more internal absorption than in planar emitters. Also, grinding domed diodes is an expensive process.
In an effort to gain the benefits of both planar and domed diodes, many manufacturers now coat planar diodes with a transparent dome of epoxy which essentially serves the same purpose of the semiconductor dome.
Physics of Light Emitting Diodes
Modern atomic physics tells us that electrons within the confines of a particular atom are allowed to occupy only discrete energy levels or bands. Energy levels between the outer two bands, the valence and conduction bands, are separated by a region called the forbidden gap. Under special conditions of doping, an electron may occupy one or more levels within the forbidden gap for relatively brief periods of time. The forbidden gap plays an important role in semiconductor light emission, since transitions from the conduction to the valence hand provide the mechanism for photon generation.
An efficient way to cause transitions between the valence and conduction bands is to pump or inject electrons into the n region of a semiconductor diode. If a sufficient number of electrons is injected, the potential barrier at the junction of the n and p regions will be overcome and current will flow through the diode. Having crossed the junction barrier, the injected electrons seek their equilibrium point and drop from their excited position in the conduction band to the valence band. In essence, the electrons drop into holes or regions in a band where there is an electron deficiency.
The act of electrons combining with holes is called recombination and results in the magic of PN junction electroluminescence or light emission. An electron falling from a high to a low level releases the energy which propelled it over the junction as heat, light, or some of each. Heat emission is accompanied by vibrations in the crystal lattice and if left uncontrolled, results in thermal destruction of the diode. Heat emission predominates in indirect band gap semiconductors such as silicon and germanium. The forbidden gap in such materials permits an electron falling from the conduction to the valence band to loiter briefly at one or more levels. Transitions between the various levels in the forbidden gap may result in either light or heat emission.
It is interesting to note that an ordinary silicon or germanium diode emits a minute quantity of infrared light when forward biased. However, light emission in such diodes is far less efficient than heat production and develop-ment of practical semiconductor light emitters followed research with direct band-gap materials.
Gallium arsenide GaAs is normally a direct band-gap material. Since electrons injected into a GaAs diode recombine with holes without pausing at intermediate levels within the forbidden gap, light emission is very efficient and production of heat is generally limited to contact resistance and bulk absorption of some of the emitted light within the semiconductor.
GaAs diodes are so efficient as light emitters that scientists chose them as candidates for early work concerning the feasibility of fabricating semiconductor lasers. In the fall of 1962, researchers at G.E., IBM, and MIT announced almost simultaneously lasers made from specially prepared GaAs diodes. In a future issue of POPULAR ELECTRONICS, the operating principles and characteristics of the diode laser will be described.
How Are They Made?
As mentioned earlier, the LED usually consists of a PN junction of one type of semiconductor, the most common being gallium arsenide. A typical diode may consist of a small wafer of positively charged (p) material in intimate electrical contact with a metal header similar to a transistor case. Before it is mounted on the header, the wafer is given a thin top layer of negatively charged (n) GaAs by either diffusion or epitaxial growth. The P portion of the finished wafer is connected to one of the wire leads of the header, resulting in a true PN junction diode.
To collect the light output from the wafer in an efficient manner, a metal can with a lens or flat window at one end is welded to the header. The can also serves as protection (see Fig. 4).

Fig. 4 - In practical applications, the LED is mounted within a metal enclosure, similar to a transistor, for mechanical protection. A transparent window permits the light to exit from one end of the enclosure.
How Are They Used?
Now that we know something about the history, physics, and mechanical construction of the LED, what are its applications other than those found in the laboratory? Especially, how can the experimenter use these tiny sources of "cold light."
An important military use of the LED is as a covert source of illumination for night vision devices. The usefulness of the Army's starlight scope is greatly enhanced by invisible infrared illumination supplied by one or more LED's. Another military use is in covert communications. The Navy, for example, has several types of infrared, line-of-sight LED voice communicators for ship-to-ship and ship-to-shore.
Recently, commercial applications of the LED have been revealed at an unprecedented rate. Several companies are mass producing tiny alpha-numeric displays consisting of arrays of LED's. These displays operate at a much lower voltage than conventional mechanical, incandescent, cathode ray, or gas-discharge displays.
One manufacturer has demonstrated a complete VOM in a probe! Using several of the new numeric displays, the futuristic unit is similar in size to a standard scope probe. Volts and ohms are read directly from the small light display. The Hamilton Watch Company will market a digital watch using LED numeric displays, and Bell Telephone is hard at work on new types of blue, green, yellow, and red LED's for use in home telephones. Infrared LED's are being used in several types of new intrusion alarms. One company even markets a complete line of LED voice communicators and burglar alarms.
One of the most unusual applications of the infrared LED is as a light source for small mobility aids for the blind. Such devices may eventually be marketed at a price comparable to most hearing aids.
An important result of all these new uses for LED's is a large drop in price. LED's are available for under $2.50 each in small quantities. This price is competitive with that of miniature, long-life indicator lamps. And of course, the LED offers sturdier packaging, a million-hour lifetime, and far less current consumption.
Only a year ago, the cheapest infrared LED retailed for $18.00. At least five manufacturers now offer GaAs LED's for under $7.50. These devices are far more efficient than visible LED's and are therefore usable in experiments in secret communications.
To sum up then, the LED is superior to the conventional filament lamp in the following ways:
1. Response time is extremely fast - most LED's operate in a fraction of a second rise and fall time . Some LED' can reach a speed of 100 MHz.
2. Because it is a solid-state device, an LED has no warm-up time, is completely free of microphonics, is just about impervious to mechanical vibration and other environmental conditions, and is usually of very small size and weight.
3. The LED light output is nearly monochromatic-far from being a laser - but close enough to have most of its light in a relatively narrow bandwidth. This makes it possible to use optical filters to reduce ambient noise.
4. The LED is a low-impedance device with forward characteristics similar to those of a conventional silicon diode and it can be driven from ordinary low-voltage supplies with conventional transistor circuitry.
Elsewhere in this issue you will find a unique construction article for a low-cost LED communicator. Other projects using LED's will appear in future issues.
More Information
For more information about commercial light emitting diodes, write to one of the following manufacturers:
- Electro·Nuclear Laboratories 115 Independence Drive Menlo Park, CA 94025
- Miniature Lamp Department General Electric Company Nela Park Cleveland, OH 44112
- Monsanto Electronic Special Products 10131 Bubb Road Cupertino, CA 95014
- Radio Corporation of America Electronic Components and Devices Harrison, NJ 07029
- Texas Instruments Semiconductor Components Division Box 5012 Dallas, TX 75222
- Fairchild Semiconductor 313 Fairchild Drive Mountain View, CA 94040
- Hewlett-Packard 1501 Page Mill Road Palo Alto, CA 94304
- Motorola Semiconductor Products Box 20912 Phoenix, AZ 85036
- Sharp Electronics Corporation 178 Commerce Road Carlstadt, NJ 07072
- An excellent booklet on LED's is sold by the General Electric Company for $2.00. Called the "Solid State Lamp Manual," it describes in detail theory, characteristics, and applications.
Posted April 19, 2023
(updated from original post
on 9/16/2011)

Copyright: 1996 - 2026 |
About RF Cafe RF Cafe began life in 1996 as "RF Tools" in an AOL screen name web space totaling 2 MB. Its primary purpose was to provide me with ready access to commonly needed formulas and reference material while performing my work as an RF system and circuit design engineer. The World Wide Web (Internet) was largely an unknown entity at the time and bandwidth was a scarce commodity. Dial-up modems blazed along at 14.4 kbps while tying up your telephone line, and a lady's voice announced "You've Got Mail" when a new message arrived... |
Copyright 1996 - 2026 All trademarks, copyrights, patents, and other rights of ownership to images
and text used on the RF Cafe website are hereby acknowledge My Hobby Website: My Daughter's Website: |