Nuclear Radiation ... Insidious Polluter
|
|
Cesium-137, iodine-131, carbon-14, plutonium-239, strontium-90, uranium-235, and the list goes on. These and other radioisotopes associated with nuclear material are the result of explosions, medical treatments, laboratory experiments, or in some cases naturally occurring deposits. Regardless of the source, most people, including me, cringe at the thought of being exposed to the insidious effects of the cell-altering energy they possess. Ionizing radiation is the dangerous type of radiation due to its ability to dislodge electrons from atoms, and in the process forming cancerous cell mutations or killing the cells altogether. Researchers in the early days of radiation discovery experienced sometimes gruesome maladies as a result of the handling isotopes. Some knowingly subjected themselves to harmful doses in order to further science. Although I couldn't find it now in a search, I recall reading about and seeing a photo of a doctor who purposely exposed one of his own hands to x-rays to determine energy levels hazardous to human skin tissue. Parts 1, 2, and 3 of this series appeared in the October, November. and December 1972 issues of Popular Electronics, respectively. See also "Nuclear Radiation ... Insidious Polluter" in the February 1972 issue. Nuclear Radiation ... Insidious Polluter
By Forest H. Belt Most people relaxed and quit worrying about fallout and the "bomb" nearly a decade ago. The Limited Test Ban Treaty (June 1963) ended atmospheric testing of nuclear weapons. Only Red China and France have exploded bombs since then with predictable radioactive clouds waft-ing across continent and ocean. Two-layer plastic radiation suits reduce exposure danger for workers entering contaminated area. Umbilical tube carries fresh air and communication wiring. On leaving area, worker simply shucks the outer suit. (Photo: US Atomic Energy Comm. Richland Operation) In spite of the "now" issues, the specter of harm from nuclear radiation is still with us. Tests of nuclear explosives continue, although most are performed underground. Nuclear power plants dot the nation. The result is that chances of radiation pollution keep mounting. Some scientific authorities say that we relaxed under false assurances. The Atomic Energy Commission keeps telling us that none of today's uses of nuclear energy pre-sent any significant danger. Yet, the evidence keeps piling up that potential radiation danger lurks in peaceful uses of nuclear power. We have accumulated more knowledge of the Hiroshima/Nagasaki survivors and their descendants. Some genetic consequences were already acknowledged. We know now that even the grandchildren of the survivors are affected. Abnormalities include birth defects, stillbirths, and heredity-related diseases. Lately, cancers began appearing in the bomb victims themselves, and at a rate greater than the national average for all of Japan. And the incidence of the disease is roughly proportional to the amount of radiation the survivors received during the five-year period following the bombings. Accelerated aging is noticeable in some victims who were exposed to fallout when they were very young. Consequences like these, turning up so long after exposure, require that we reevaluate our thinking. What illnesses will workers near radioactivity contract? What effects will they have on their offspring in the future. Proponents of mare nuclear power put forth a threshold theory which assumes that there is some small radiation dosage below which effects are zero. This concept is based mainly an the lack of any immediate symptoms when the radiation dose is very low. But evidence gathers far the linear theory, that radiation-related diseases and genetic aberrations increase in proportion to the radiation one accumulates, even to the most minute dosage. This means that radiation might have a cumulative effect. It is possible then that future generations well may carry the burden of any radiation carelessness now.
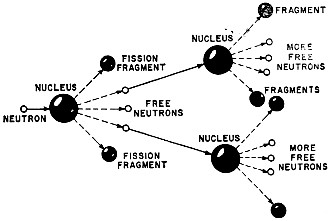
Nuclear fission occurs when a high-speed neutron splits nucleus of uranium or plutonium. Tremendous energy is released and other neutrons are knocked loose. If released energy is sufficient, chain reaction can occur. Radioactive Materials. If neutrons forced out of atoms as a result of nuclear reaction reach the air, they can mix with nitrogen atoms and form carbon 14. This radioactive nuclide has an extremely long life and can be very dangerous to man. Obviously, then, in employing nuclear reactors for peacetime applications, neutron escapes must be prevented. The decay of uranium-235 also forms variants called isotopes. If the isotopes get out of a reactor, they can carry radiation to us in various ways, mainly by concentrating in our food and water. Strantium-90, for example, settles to earth where grass absorbs and concentrates it in its blades. Cows grazing on the contaminated grass increase the concentration in their milk. Anyone who drinks the milk gets a hefty dose of the beta rays given off by the isotope. Other nuclides collect in other foods, animals and fish. Many of these nuclides, in addition to giving off beta rays, are contaminated with alpha or gamma radiation. Beta radiation is not strong. Skin, muscle, and other tissue help to block it when the body is externally exposed. Taken internally, however, strantium-90 settles in bone and muscle where beta rays freely emanate for more than 25 years. This is an open invitation to leukemia and cancer. Editor's Note: There are many benefits to be derived from the peaceful use of nuclear energy, especially in the fields of power generation and medicine. Although this article points out the dangers involved, we can take advantage of the benefits and minimize the hazards by enlisting the aid of electronics and other safeguards in control and monitoring. Those involved with the applications of nuclear energy are well aware of the problems in working with such a potent force. Hence, safe operating practices are observed to keep all hazards to an absolute minimum. The isotope cesium-137 also goes to muscles and bones. Beta and gamma rays from it endure beyond the nuclide's 33-year half life. (Note: Radionuclides are considered to be ineffective after half their atoms have decayed, although some of their rays remain potent for a longer time.) Gamma rays are intensely more penetrating than are beta rays. They can destroy body cells from relatively great distances. Iodine-131 gathers in thyroid and salivary glands. This isotope has a half-life of only eight days, but gamma and beta rays from it are energetic. They damage sensitive throat cells and trigger thyroid cancer. Plutonium-239 is the most hazardous byproduct of uranium-235 fission. Its half life is 24,100 years. It is a deadly source of alpha and gamma rays that are so powerful that they still produce lung cancers when the isotope has aged 200,000 years! Exposure to fresh plutonium-239, even briefly, multiplies cancer susceptibility a thousand-fold. Ionizing radiation produced by atomic decay attacks body cells. Radioactive particles rip electrons out of atoms in cells. Sometimes the shock kills the cell, sometimes it merely fouls up operation. Damaging a body cell sets the stage for cancer and other diseases. Chromosome damage in the reproductive cells leaves consequences that may linger for generations. Some cell damage self heals. Some damage is irreparable. The body often replaces dead cells, which leads some experts to contend that there is a threshold level below which radiation will do no somatic or genetic damage. True, below certain levels no immediate clinical symptoms appear, but radiation doses accumulate and eventually produce effects that are noticeable. Nuclear test blast in Nevada near Los Alamos lab had nearby tower that held electronic instruments to measure seismic shock, radiation, etc. (Photo Los Alamos Scientific Lab) Typical Radiation Doses. We cannot see, hear, taste, or feel radiation, but it is always around us. We get yearly background dosages of about 125 millirads - more if we live at high altitudes. (A rad is a measure of radiation standing for Radiation Absorbed Dose and is roughly equal to a Roentgen, or rem, which is a technical unit of radiation. The rad is abbreviated as the letter "r.") An average of 50 mr of annual background dosage is due to cosmic radiation from high-energy protons originating in outer space. Another 50 mr or so comes from potassium-40, thorium, and uranium in the air, soil, and buildings. And about 25 mr comes from inside our bodies. We ingest through breathing, eating, and drinking traces of thorium, potassium-40, carbon-14, and tritium; and there is no way to avoid them. Whenever we have an X-ray picture taken, we get a dose of radiation. The younger we are, the more damage X-radiation is likely to cause. A study by Dr. Alice Stewart of Great Britain - confirmed by Dr. Brian MacMahon of Harvard - relates X-rays during pregnancy to a rise of cancer and leukemia in children age 10 years and younger. X-rays are, nevertheless, beneficial diagnostic tools in medicine. The American College of Radiology, however, advocates reduction of per-photo dosage; this will help. X-rays, cobalt-60, and other radioactive isotopes also have high therapeutic value. But in spite of their obvious beneficial qualities, they remain sources of hazardous radiation. Safety guidelines have been set up by the Atomic Energy Commission, with aid provided by the Federal Radiation Council, the National Committee on Radiation Protection and Measurement, and the International Commission on Radiological Protection. The guideline doses are represented as "safe" exposures. But in light of recent data and experience, some scientists in the nuclear and health fields dispute their safety. Articulated mechanical arms and hands let chemist work with radiopharmaceuticals in safety behind concrete wall and sheets of heavily leaded glass. (Photo: USAEC) The guidelines suggest that the average dosage for the population at large be less than 170 mr/yr, not counting natural back-ground radiation. Anyone person should receive no more than 500 mr/yr, say the guidelines. Individuals in radiation-associated jobs are permitted an "acceptable" dosage of 5000 mr/yr - 40 times the background level. Medical Physicists John W. Gofman and Arthur R. Tamplin of the Lawrence Radiation Laboratory in California make a strong case that even 170 mr/yr can induce an extra 32,000 cancer (plus leukemia) cases annually, representing a 10 percent rise over the usual average. Also, genetic effects might add 150,000 deaths and even more deformities and diseases. Naturally, nuclear industries take elaborate precautions to maintain exposure below the AEC maximum. Shielding and special suits are used to block nuclear rays. Remote control handling keeps operators away from critical materials. Instruments monitor radiation levels where contamination might exist or develop. Some cities, like New York, monitor nuclear radiation level constantly. Film badges check the daily and weekly rad dosages of workers who risk exposure. But for the ordinary citizen, the only real safety lies in prevention. Fission vs. Fusion Power. There is currently a big controversy raging with reference to nuclear-electric power plants. The plants may look clean and harmless, but their reactors are sources of radioactive discharges into the surrounding environment. There is also the possibility that accidents caused by man or nature (freak tornadoes, floods, earthquakes, etc.) could spread radiation for miles. Hauling and storing nuclear leftovers also present pollution hazards. This particular problem worries Kansans around the Lyons salt mines where nuclear waste is being dumped. And there are dangers near plants where reactor fuels are processed. Many worried citizens want no more nuclear power plants built unless they release no contaminants at all. California, Oregon, Minnesota, and New York City have even legislated moratoriums that are still in the courts. Meanwhile, power companies state that we must convert to nuclear power because we have enough fossil fuel to carry us through only 200 more years. This is a poor argument considering that we have only 75 years' worth of uranium at our present rate of consumption. Emergency radiation team members at AEC's Hanford Works show how Geiger and scintillation counters are used to measure radioactive contamination should it get out of control of users of radioactive isotopes. (Photo: Hanford Oregon, Atomic Products Operation) Fast-breeder reactors can develop large quantities of a plutonium-239 byproduct that can be fissioned into other reactors. But plutonium is formidable to store and transport. Fusion, the thermonuclear reaction used in the hydrogen bomb, offers an alternative. Its deuterium fuel, a heavy isotope of hydrogen, is abundant and relatively inexpensive. A little generates a lot of power, and known accessible reserves could last us 50,000 years. But most promising, for equivalent energy released, a fusion reaction produces about a million times less radioactivity than does a fission reaction. The problem is that fusion reaction is not easy to produce or control. We have machines that can take care of both jobs on a limited basis, but none is ready for commercial exploitation, nor are they likely to be for a decade or more. The chief radiation danger from fusion is high-energy neutrons. As already stated, neutrons form carbon-14 in air. Also, neutrons wear out the protective vanadium allays used in reactors. Tritium, produced within the reactor, has a half life of 12.5 years and emits strong beta rays. So, it must be kept from contaminating air or water. A deuterium-helium thermo nuclear reaction can make electricity directly. The method is being worked out at the Lawrence Radiation Laboratory. A plasma is confined in a straight tube called a "magnetic mirror machine." The reaction spews electrons out of the tube into an expansion chamber. Collector electrodes gather positive charges and a charge separator picks up the electrons. Output terminals feed the direct current to whatever load is provided. Fusion reaction is a relatively "clean" method of generating power, but mechanical problems in employing it on a universal scale are still far from resolved. Consequently, we are stuck with fission reactors to produce energy; so, we are also stuck with the dangers they present - unless legislation eliminates the use of nuclear power plants altogether. A Growing Tide. We still test nuclear weapons in spite of the April 1970 Treaty an Nonproliferation of Nuclear Weapons and our present Strategic Arms Limitation Talks. Controversial underground tests continue in Colorado and Nevada. And, in early November, a giant 5-megaton thermonuclear device for our anti-ballistic missiles was tested a mile beneath the ground on Amchitka Island. Neither earthquakes nor tidal waves were triggered and the AEC states that no radiation escaped into the atmosphere. Missile submarines such as the USS George Washington and ships like the carrier USS Enterprise carry fusion reactors for propulsion, No reactor is entirely "clean": so, only "safe" doses of radiation reach the men who run our nuclear fleet. Portable radiological survey instruments built by private industry for Oak Ridge National Laboratory. Individual meters are for neutrons, beta and gamma rays, and alpha particles. (Photo: Oak Ridge National Laboratory) The National Aeronautics and Space Administration has a nuclear-powered rocket called NERVA (Nuclear Engine for Rocket Vehicle Application). Nuclear propulsion could considerably shorten trip time between earth and moon. Astronauts already get nearly 1000 mr of radiation exposure on an Apollo trip, mostly from traversing the Van Allen radiation belts. A quicker trip through the belts may balance out the effects of radiation from the engine. A new nuclear-electric racket develops only a few pounds of thrust. However, it operates continuously over a long period of time; so, deep-space vehicles could build up extreme velocities aver the long haul. Systems for Nuclear Auxiliary Power, or SNAP, thermoelectric generators already make electricity for scientific experiments on the moon. Larger versions might serve earth needs. Our SNAP-27 uses plutonium-239, but scientists are working on safer versions. The Big Ditch. Massive nuclear explosives offer engineering promise. But radiation problems hamper the use of such power. A detonation in 1967, called "Gasbuggy," attempted to boost gas output of a well in New Mexico. Rulison, a similar project in Colorado, took place in 1969. Unfortunately, the gas released is dangerous; no matter whether fission or fusion explosives were used, tritium or krypton-85 in the gas exposes customers to radiation. Decontamination is too costly or impassible. Tests in oil fields also flapped; the blasts left too much tritium in the ail output. The most exciting excavation proposal to date is the large sea-level canal to be cut across Central America to replace the soon-outmoded Panama Canal. Thermonuclear blasts appear to be the only economical means for such difficult digging chores - but not without problems. Nuclear tests have been detonated above-ground very little since 1963. Engineers are not sure how to deploy the explosives to achieve the desired results. Seismic shock could tear up nearby towns, and the acoustic shock wave has its own dangers. But worse still is the fact that scientists simply cannot produce a radiation-free nuclear explosion. The cleanest fusion explosives still dump neutrons and tritium into the atmosphere. The new canal is therefore stymied. So are other excavation projects. Truly safe peacetime nuclear technology awaits fission or fusion that does not cause radiation pollution.
Posted May 22, 2018 |
|

 Electronics helps monitor and control contamination
you can't see, hear, taste, smell, or feel
Electronics helps monitor and control contamination
you can't see, hear, taste, smell, or feel