Nuclear Radiation & Detection - Types of Radioactivity
|
|
This is an instance where I scanned and posted later parts of a multi-month article because I had not yet purchased the edition with the first in the series, but I have it now. Author J.G. Ello, of the Radiation Measurements and Instrumentation Electronics Division of Argonne National Laboratory, introduces the physics of nuclear radiation along with the genesis and evolution of nuclear technology. Alpha, beta, and gamma radiation is explained, as is the decay phenomenon and energy levels. It is all at an introductory level, and from a classical Bohr model perspective. If you are new to nuclear physics, these four articles are a good starting place. Parts 1, 2, and 3 of this series appeared in the October, November. and December 1972 issues of Popular Electronics, respectively. See also "Nuclear Radiation ... Insidious Polluter" in the February 1972 issue. Nuclear Radiation & Detection - Part 1: Types of Radioactivity
By J. G. Ello, Radiation Measurements and Instrumentation Electronics Division, Argonne National Laboratory About 30 years ago in Chicago, scientists built and successfully operated the world's first nuclear reactor. Basically speaking, the scientists had an atomic bomb detonation under control. Since then, the atomic age has progressed into many fields throughout the world. Radioactivity, for example, is used in prospecting for ore, coal, and oil deposits. Space satellites and deep sea research use radioactivity to provide electrical power; hospitals use it for tracers, cancer research, and treatment; and its tracers are used in agriculture for fertilizer improvements. Radioactivity is also used in crime detection to identify pollution and determine the age and history of various materials. Currently, radiation is being used in industry to determine the density and thickness of various materials. Sterilization and pasteurization of food and insect control are other areas in which radiation is being used. Perhaps the greatest potential use of all is in the generation of electrical power.
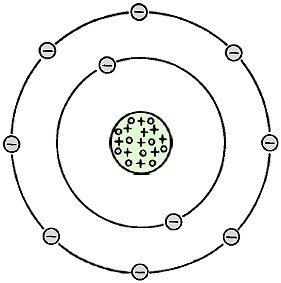
Fig. 1 - Makeup of a stable neon atom - atomic number 10. Radioactivity is not new on earth. It has been around ever since the earth was formed, even though we are only now beginning to put it to use. In 1895 when radioactivity was discovered, W. C. Roentgen in Germany was experimenting with electrical discharges through evacuated glass tubes when he discovered the rays coming from the tubes were able to cloud photographic plates and ionize gases. In his notes, he referred to these rays as X rays. Following the discovery of X rays, the French physicist, H. Becquerel, found that a piece of uranium would also darken photographic plates. In 1898, Pierre and Marie Curie discovered that other elements which they named radium and polonium also emitted rays that could ionize gases and darken photographic plates. They called this phenomenon "radioactivity." In 1899, while studying in England, New Zealand physicist E. Rutherford found that, when radioactivity passed through thin sheets of aluminum, its ionizing power was reduced. From this, he concluded that the radiation emitted from uranium was up made of two types which he called alpha and beta radiation. In 1900, a third type of radiation emitted from uranium was discovered by P. Villard. Finding it to be similar to X rays, he named it gamma rays. Elements For 2000 years, dating from the fifth century B.C., it was believed that all matter was made up of four elements - fire, air, water, and earth. For many centuries, ancient alchemists attempted, without results, to change base metals into gold. With present technology, however, it is possible to transmute or change one element into another. It was not until about 1789 that Lavoisier established that an element was a substance containing only one type of matter which could not be split into anything simpler. He listed about 30 such elementary substances of which about 20 are to this day regarded as elements. By 1819, the number of elements was increased to 50. Presently, there are 92 known natural elements. Among these are gold, silver, nickel, lead, and uranium. With modern technology, elements can be produced by transmutation and disintegration. These include americium, curium, berkelium, plutonium, and neptunium (the latter two, a result of the atomic bomb). The current count of elements, including those which are man-made, is 103, by now possibly 105. Radioactive Atoms Since radioactivity is concerned with the disintegration or decay of an atom, it is worthwhile to take a close look at the atom. The atom is held together by a force of attraction. It consists of a nucleus and orbiting electrons. In a very basic sense, the atom is similar in structure to the Solar System with the sun as the nucleus and the planets as the orbiting electrons. The planets in our Solar System are held together by gravitational force. The atom, however, is held together by an electrical force. A stable neon atom is shown schematically in Fig. 1. In its nucleus are 10 protons and 10 neutrons. Orbiting around the nucleus are 10 electrons, two of which are in the inner ring and eight in the outer ring. The protons carry a positive (+) charge, the electrons a negative (-) charge, and the neutrons are neutral, having no (0) electrical charge. Similarly, a uranium atom consists of a nucleus with protons and neutrons and a comparable amount of electrons (92) in seven rings. Uranium, however, is not a stable atom. It will decay until it eventually becomes lead. When uranium, radium, or plutonium decay, they give off particles and rays - radiation. It takes one pound of uranium about 4.5 billion years to decay to one-half pound and another 4.5 billion years to decay to a quarter pound, and so on. The rate of decay for radioactive elements is based on the elements' half-lives, which vary from element to element. For example, the half-life of radium is about 1600 years, plutonium about 24,000 years. Other radioactive elements may decay in less than a millionth of a second. Alpha, Beta & Gamma Radiation Fig. 2 - Courses in a magnetic field (top) and penetrating powers (bottom) of alpha, beta, and gamma radiation. From the very beginning, man has been bombarded by cosmic radiation from the sun and the distant stars. Owing to the atmospheric shield with which nature has provided us, the cosmic rays are reduced to intensities we consider harmless. The natural radiation with which we must contend as part of our environment is defined as alpha, beta, and gamma types. How the three differ in their penetrating power and their course in a magnetic field is shown in Fig. 2. It has been stated that radium and uranium each emit all three types of radiation as they decay. However, it should be understood that not all radioactive elements give off all three types of radiation. The alpha type, first described as a "ray" but later found to be a particle, is a helium atom with a positive charge as a result of having lost two of its electrons. The alpha particle is ejected from a radioactive atom at a velocity about 1/20 that of light. The alpha particle has a high kinetic energy potential as a result of its high speed and large mass. It loses a little of its energy each time it collides with another atom. The energy is dissipated each time it knocks an electron out of an atom with which it collides; during the process ions are formed. Although the alpha particle can travel through 1-3 in. of air, it cannot penetrate a few sheets of newspaper. Too, being a positively charged particle, it can be deflected in a magnetic field. The beta particle, like the alpha particle, is also emitted from the nucleus. But the beta particle can travel several hundred times farther than the alpha particle and at a velocity approaching that of light. It can be absorbed by a 1/4 in. thick sheet of Lucite. The beta particle has the same mass and charge as an electron, and compared to an alpha particle, it has 7500 times less mass. Therefore, a beta particle with the same energy as that of an alpha particle will travel much farther and faster before it is completely absorbed by collisions with other atoms. Similar to the alpha particle, the beta particle leaves ions in its wake. Through the use of electronic equipment, these ions (in both cases) provide us with much useful information. The third type of radioactivity is known as the gamma ray. Unlike alpha and beta particles, the gamma is a ray - not a particle. It is an electromagnetic wave, similar in behavior to radio waves and microwaves, ultraviolet waves, and X rays. To be more precise, gamma rays are X rays of very high frequency and very high penetrating power. While alpha particles are absorbed by a few sheets of newspaper and beta particles by a 34 in. thick sheet of Lucite, it may take several inches of lead to absorb radium's gamma rays and several feet of concrete to absorb gamma rays produced by nuclear power reactors. Since gamma rays are not particles, they are unaffected by a magnetic field. Gamma rays are emitted from the nucleus of an atom when an excess of energy remains after the ejection of an alpha or beta particle. Unlike alpha and beta particles which produce ions in their wake, gamma and X rays produce ionization through a secondary effect. When a gamma ray enters an atom, it will more likely dislodge an electron which will, in turn, ionize other atoms by dislodging other electrons in its path. As we shall see in Part 2 in this series, the secondary ions are collected in a detector and the count indicated on a radiological survey meter. More To Come. We have come to the end of Part I in this three-part series. In this installment we have presented and discussed the various types of radioactivity. Parts 2 and 3, which will deal with Radiation Detectors and Radiological Survey Meters, will be presented in the November and December issues.
Posted September 25, 2019 |
|