Recent Developments in Battery Design
|
|
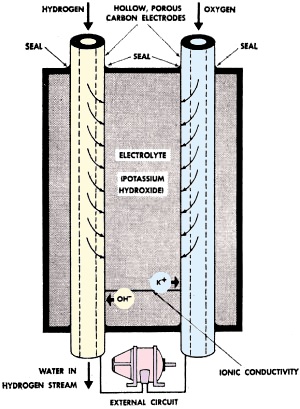
"Mr. Scott, prepare to load the promethium batteries into the dilithium crystal chamber." OK, I made up the promethium batteries part, but you might not have suspected it. Back in the mid to late 1950s, atomic batteries were seriously thought to potentially (no pun intended) be a futuristic source of energy storage and generation. The concept worked by having beta particles from promethium decay impinge on silicon photodetectors, and having that be the source of power. That's almost as Rube Goldberg−ish as having a gasoline engine drive an electric generator to power a motor for automotive locomotion. Oh, wait, that describes the Chevy Volt. Note that in 1959, nickel cadmium (NiCad) batteries were just coming into commercial use, and the author envisions a day when they might be used for portable power tools and flashlights. At least he was right about that one. See the 1957 edition of Time magazine with an article titled, "Science: New Atomic Battery." Recent Developments in Battery Design Two soldiers at a front-line observation post crouch behind a strange-looking device that looks like a spotlight: As they wait in the darkness, instead of straining their eyes to discern enemy movements, their attention is riveted to the spotlight-like device. Capable of spotting a single enemy soldier a half-mile away in total darkness, this is the Army's new "Silent Sentry" mobile radar set. Atomic batteries, fuel cells, and other new battery types hold great promise for the future by Saunder Harris, WINXL Under battle conditions quiet operation of the Silent Sentry could be a matter of life or death; therefore, a noiseless source of power for the unit is essential. Obviously, here is a job tailor-made for batteries. But which type should be used? Of a whole parade of new batteries, the Army has chosen one of the newest-the little-known fuel cell-to power the Silent Sentry. The fuel cell is only one of the new battery types which are today proving their worth. Already on the commercial market are the tiny, power-packed mercury cell and the rechargeable nickel-cadmium battery. Before long, the fuel cell, too, will be available to private citizens. And the most fascinating development in batteries - the amazing promethium cell which is powered by energy from the atom itself - is now be-ing tested and refined in research laboratories in this country. Let's examine these "wonder" batteries. First, since it's one of the newest, we'll take a look at the fuel cell, the power source of the Silent Sentry. Fuel Cell The scientist who did much of the research on the fuel cell, Dr. Karl Kordesch, examines an electrode used in the cell. The most interesting characteristic of the fuel cell is that, unlike conventional batteries, it never becomes exhausted. Since it produces electrical current from the electrochemical reaction which takes place when oxygen and hydrogen are combined, the fuel cell itself remains usable as long as the "fuel" - oxygen and hydrogen - is supplied. Operation of the fuel cell is diagrammed in Fig. 1. Oxygen and hydrogen enter the cell through two hollow carbon electrodes. Since these electrodes are porous, the gases rapidly diffuse to the outer surface of the electrodes where they come into contact with the electrolyte, a solution of potassium hydroxide. The chemical reaction which takes place releases electrons from the hydrogen electrode which flow through the external circuit and are returned at the oxygen electrode. It is this flow of electrons which provides the electric current. Water, a by-product of the reaction, is passed from the cell in the hydrogen stream. One fuel cell can produce only about one volt. Any required voltage may be attained, however, by simply connecting the cells together in series. As with ordinary dry cells, the amount of current which can be drawn from a fuel cell is a function of its physical size. Thus, by varying the number and size of the cells, many variations of voltage and current can be obtained. Fuel cells have been operating eight hours a day, five days a week, for over a year with no sign of deterioration.
Fig. 1 - Operation of the fuel cell. Electron flow from hydrogen electrode to oxygen electrode provides electric current.
Nickel-cadmium and mercury batteries such as these are already on the market.
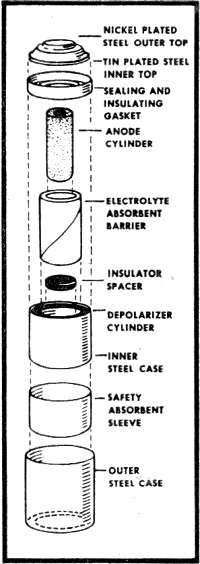
Fig. 2 - Exploded view of mercury battery (courtesy National Carbon Company)
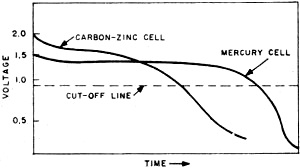
Fig. 3 - Comparison of discharge curves of mercury cell and carbon-zinc cell. It is readily seen that the mercury cell enjoys a much longer useful service life.
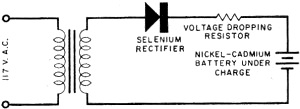
Fig. 4. Typical simple rectifier circuit employed to reactivate rechargeable batteries.
Fig. 5 - Promethium cell composed of center layer of promethium and phosphor with outer layers made up of photocells. It is very possible that the fuel cell will be the practical means of putting both nuclear energy and solar energy to use. At present, one of the big difficulties involved in using the energy of the sun is in storing its power for future use. Now, during the sunlight hours, the sun's energy could be used to decompose water, producing both hydrogen and oxygen for later use in fuel cells. In the same manner, where nuclear reactors are used as heat sources in steam generating plants, the nuclear energy decomposes water. Instead of this being a disadvantage, as it has been, this process can now be the means of producing the necessary hydrogen and oxygen for fuel cell operation. Mercury Batteries Wherever long life and lots of punch must be jammed into a small package, the mercury battery has come into wide use. This battery was developed primarily for hearing aids and other ultra-miniature equipment. An exploded view of a typical mercury battery is shown in Fig. 2. The materials used in its construction are high-purity zinc powder for the anode, mercuric oxide and carbon for the cathode, and potassium hydroxide as the electrolyte. The mercury cell develops an open-circuit voltage of approximately 1.35 volts. Figure 3 illustrates the difference in performance between the mercury cell and the standard carbon-zinc cell. It can be readily seen that the voltage of the mercury cell remains constant over a longer period of time than does the carbon-zinc cell. For general use, the mercury battery is very expensive. The size "D" mercury battery (flashlight size) costs about $2.50 as compared with a price of 20 cents for an ordinary carbon-zinc "D" cell. This high cost is due to the expensive materials used in construction. Mercury batteries became financially practical only when devices such as hearing aids were designed to use them. Nickel-Cadmium Battery Along with other dry cells, the mercury battery suffers from one big disadvantage. It cannot be recharged. However, rechargeable nickel-cadmium batteries are just coming into popular use today in rechargeable flashlights, radios and electric razors. Tomorrow they may be used to power TV sets, portable electric drills, and perhaps even your car. After conventional dry cells have been used awhile, the action of the cell is gradually choked off by a gas which is developed in the cell. In the nickel-cadmium battery, the recharging process converts this gas back into a liquid, thus reactivating the cell. Recharging is accomplished by plugging the battery unit into the house power line. Figure 4 shows a typical half-wave rectifying circuit used for this purpose. In most cases, the rectifier is built into the same unit as the battery. Nickel-cadmium cells can't give you something for nothing, however. Rechargeable cells cost more, and give less energy per charge than an ordinary carbon-zinc cell. For example, a nickel-cadmium flash-light cell costs about $2.75 as against 20 cents for a comparable carbon-zinc cell. But it can be charged over and over. Even more expensive than the nickel-cadmium battery is its highly refined "cousin," the silver-cadmium battery. This battery enjoys all the advantages of the nickel-cadmium design, and, in addition. offers higher output at one-half to one-third its size and weight. Since the silver-cadmium battery is quite costly, its greatest application so far has been in rockets. missiles, and satellites. Atomic Batteries Much misleading publicity has surrounded the atomic battery. In spite of newspaper reports which would lead you to believe that an atomic powered radio is just around the corner this is not the case. As one battery engineer put it, "there are a great many problems to be solved before atomic batteries are brought out of the laboratories and put into your home." One objection to the atomic battery is its potential danger. How would you like to have a little package of radioactivity around the house where it might be broken into by youngsters with a yen for experiments? Another objection is cost; at this time, the material which goes into such batteries is extremely expensive. The batteries we have working for us now do a good job at reasonable cost and the idea of replacing them with atomic batteries might be novel but cannot be considered practical at present. However, laboratory research continues on the atomic battery. Radioactive promethium - promethium is a by-product of uranium fission - is the power source. It is valuable because it emits large amounts of beta rays (actually electrons) over its 2 1/2-year half-life. These beta rays can be tapped as a source of power. Alpha a gamma rays are emitted only in small quantities. The actual size of the promethium and its shielding is about that of a penny Figure 5 shows a typical promethium cell in cross section. The center layer is a mixture of promethium and phosphor. Small photocells compose the outer layers. When the promethium gives off beta rays, they strike the phosphor with great force. The phosphor then lights up in much the manner as your TV screen does when the electron stream of the cathode-ray tube hits it. This light is then converted to electrical energy by the two outer layers of photocells. Output of the promethium cell is small - actually less than one-millionth of the electrical power used by a 40-watt bulb. It does give off power, though, and the power comes from atomic radiation. Considering its early stage of development, the promethium cell shows great promise. Any discussion of batteries must necessarily lead us back to the fact that the carbon-zinc cell is still battery king of the present. And in the distant future, when you send one of the kids down to the Lunar Hardware on the Moon to pick up a battery for your flashlight, chances are you will end up with our old friend, the carbon-zinc cell.
Posted October 6, 2022 |
|