|
These units are for general physical quantities not specifically related to electricity
and magnetism. Below is a much more extensive list derived from the NIST website.
Here is a list of physical constants.
| Enthalpy |
joule |
H |
| Entropy |
joule/Kelvin |
S |
| Heat capacity |
joule/Kelvin |
C |
| Internal energy |
joule |
U |
| Luminous intensity |
candela |
I |
| Quantity of heat |
joule |
Q |
| Radiant energy |
joule |
W |
| Radiant intensity |
watt/steradian |
I |
| Radiant power (flux) |
watt |
P |
| Sound intensity |
watt/meter |
I |
| Sound energy flux |
watt |
W |
| Specific heat capacity |
joule/(kilogram * Kelvin) |
c |
| Speed of sound |
meter/second |
n |
| Thermal conductivity |
watt/(meter * Kelvin) |
l |
| Time Constant |
s |
t |
Here is a much greater list of units from the
NIST Reference on
Constants, Units, and Uncertainty. This information is in the public domain.
SI Base Units
The SI is founded on seven SI base units for seven base quantities
assumed to be mutually independent, as given in Table 1.
Table 1. SI base units
| length |
meter |
m |
| mass |
kilogram |
kg |
| time |
second |
s |
| electric current |
ampere |
A |
| thermodynamic temperature |
kelvin |
K |
| amount of substance |
mole |
mol |
| luminous intensity |
candela |
cd |
For detailed information on the SI base units, see
Definitions of the SI base
units and their Historical context.
SI derived units
Other quantities, called derived quantities, are defined in terms of
the seven base quantities via a system of quantity equations. The SI derived
units for these derived quantities are obtained from these equations and the
seven SI base units. Examples of such SI derived units are given in Table 2, where
it should be noted that the symbol 1 for quantities of dimension 1 such as mass
fraction is generally omitted.
Table 2. Examples of SI derived units
| area |
square meter |
m2 |
| volume |
cubic meter |
m3 |
| speed, velocity |
meter per second |
m/s |
| acceleration |
meter per second squared |
m/s2 |
| wave number |
reciprocal meter |
m-1 |
| mass density |
kilogram per cubic meter |
kg/m3 |
| specific volume |
cubic meter per kilogram |
m3/kg |
| current density |
ampere per square meter |
A/m2 |
| magnetic field strength |
ampere per meter |
A/m |
| amount-of-substance concentration |
mole per cubic meter |
mol/m3 |
| luminance |
candela per square meter |
cd/m2 |
| mass fraction |
kilogram per kilogram, which may be represented
by the number 1 |
kg/kg = 1 |
For ease of understanding and convenience, 22 SI derived units have been given
special names and symbols, as shown in Table 3.
Table 3. SI derived units with special names and symbols
| plane angle |
radian (a) |
rad |
- |
m·m-1 = 1 (b) |
| solid angle |
steradian (a) |
sr (c) |
- |
m2·m-2 = 1 (b) |
| frequency |
hertz |
Hz |
- |
s-1 |
| force |
newton |
N |
- |
m·kg·s-2 |
| pressure, stress |
pascal |
Pa |
N/m2 |
m-1·kg·s-2 |
| energy, work, quantity of heat |
joule |
J |
N·m |
m2·kg·s-2 |
| power, radiant flux |
watt |
W |
J/s |
m2·kg·s-3 |
| electric charge, quantity of electricity |
coulomb |
C |
- |
s·A |
| electric potential difference, electromotive force |
volt |
V |
W/A |
m2·kg·s-3·A-1 |
| capacitance |
farad |
F |
C/V |
m-2·kg-1·s4·A2 |
| electric resistance |
ohm |
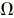 |
V/A |
m2·kg·s-3·A-2 |
| electric conductance |
siemens |
S |
A/V |
m-2·kg-1·s3·A2 |
| magnetic flux |
weber |
Wb |
V·s |
m2·kg·s-2·A-1 |
| magnetic flux density |
tesla |
T |
Wb/m2 |
kg·s-2·A-1 |
| inductance |
henry |
H |
Wb/A |
m2·kg·s-2·A-2 |
| Celsius temperature |
degree Celsius |
°C |
- |
K |
| luminous flux |
lumen |
lm |
cd·sr (c) |
m2·m-2·cd = cd |
| illuminance |
lux |
lx |
lm/m2 |
m2·m-4·cd = m-2·cd |
| activity (of a radionuclide) |
becquerel |
Bq |
- |
s-1 |
| absorbed dose, specific energy (imparted), kerma |
gray |
Gy |
J/kg |
m2·s-2 |
| dose equivalent (d) |
sievert |
Sv |
J/kg |
m2·s-2 |
| catalytic activity |
katal |
kat |
|
s-1·mol |
|
(a) The radian and steradian may be used advantageously in expressions
for derived units to distinguish between quantities of a different nature but of
the same dimension; some examples are given in Table 4.
(b) In practice, the symbols rad and sr are used where appropriate,
but the derived unit "1" is generally omitted.
(c) In photometry, the unit name steradian and the unit symbol sr
are usually retained in expressions for derived units.
(d) Other quantities expressed in sieverts are ambient dose equivalent,
directional dose equivalent, personal dose equivalent, and organ equivalent dose.
|
For a graphical illustration of how the 22 derived units with special names and
symbols given in Table 3 are related to the seven SI base units, see
relationships among SI
units.
Note on degree Celsius. The derived unit in Table 3 with the
special name degree Celsius and special symbol °C deserves comment. Because of the
way temperature scales used to be defined, it remains common practice to express
a thermodynamic temperature, symbol T, in terms of its difference from
the reference temperature T0 = 273.15 K, the ice point. This
temperature difference is called a Celsius temperature, symbol t, and is
defined by the quantity equation
t= T- T0.
The unit of Celsius temperature is the degree Celsius, symbol °C. The numerical
value of a Celsius temperature t expressed in degrees Celsius is given
by
t/°C = T/K - 273.15.
It follows from the definition of t that the degree Celsius is equal
in magnitude to the kelvin, which in turn implies that the numerical value of a
given temperature difference or temperature interval whose value is expressed in
the unit degree Celsius (°C) is equal to the numerical value of the same difference
or interval when its value is expressed in the unit kelvin (K). Thus, temperature
differences or temperature intervals may be expressed in either the degree Celsius
or the kelvin using the same numerical value. For example, the Celsius temperature
difference 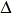 t and the
thermodynamic temperature difference t and the
thermodynamic temperature difference 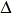 T between
the melting point of gallium and the triple point of water may be written as T between
the melting point of gallium and the triple point of water may be written as
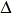 t = 29.7546 °C = t = 29.7546 °C = 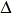 T = 29.7546
K. T = 29.7546
K.
The special names and symbols of the 22 SI derived units with special names and
symbols given in Table 3 may themselves be included in the names and symbols of
other SI derived units, as shown in Table 4.
Table 4. Examples of SI derived units whose names and symbols include
SI derived units with special names and symbols
| dynamic viscosity |
pascal second |
Pa·s |
| moment of force |
newton meter |
N·m |
| surface tension |
newton per meter |
N/m |
| angular velocity |
radian per second |
rad/s |
| angular acceleration |
radian per second squared |
rad/s2 |
| heat flux density, irradiance |
watt per square meter |
W/m2 |
| heat capacity, entropy |
joule per kelvin |
J/K |
| specific heat capacity, specific entropy |
joule per kilogram kelvin |
J/(kg·K) |
| specific energy |
joule per kilogram |
J/kg |
| thermal conductivity |
watt per meter kelvin |
W/(m·K) |
| energy density |
joule per cubic meter |
J/m3 |
| electric field strength |
volt per meter |
V/m |
| electric charge density |
coulomb per cubic meter |
C/m3 |
| electric flux density |
coulomb per square meter |
C/m2 |
| permittivity |
farad per meter |
F/m |
| permeability |
henry per meter |
H/m |
| molar energy |
joule per mole |
J/mol |
| molar entropy, molar heat capacity |
joule per mole kelvin |
J/(mol·K) |
exposure (x and 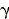 rays) rays) |
coulomb per kilogram |
C/kg |
| absorbed dose rate |
gray per second |
Gy/s |
| radiant intensity |
watt per steradian |
W/sr |
| radiance |
watt per square meter steradian |
W/(m2·sr) |
| catalytic (activity) concentration |
katal per cubic meter |
kat/m3 |
|